Experiment #14: Polymers
I've been Slimed
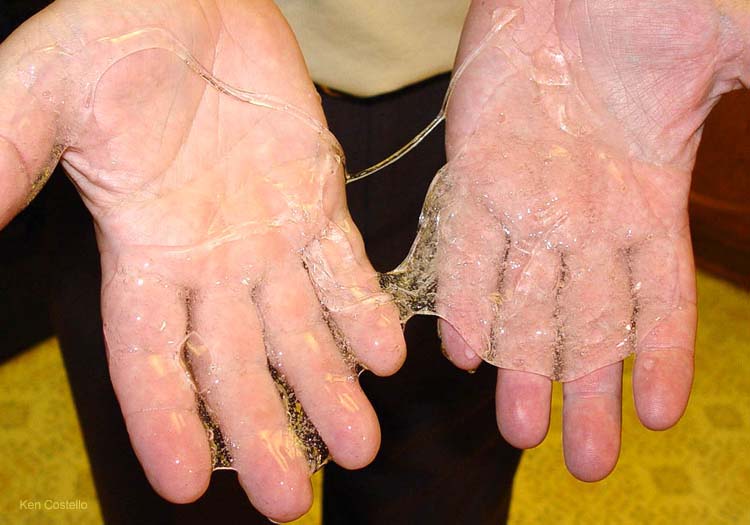

The Ghostbusters made slime famous with "I've been slimed" line from Bill Murray and the scenes of ectoplasm (slime) used throughout the movie.

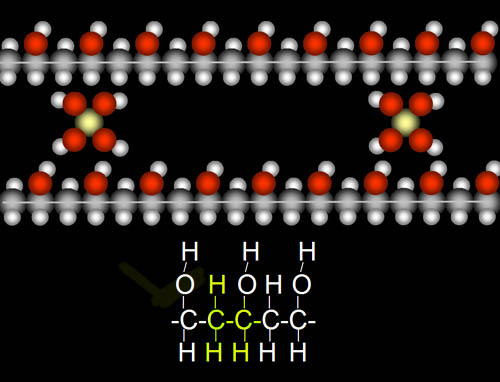
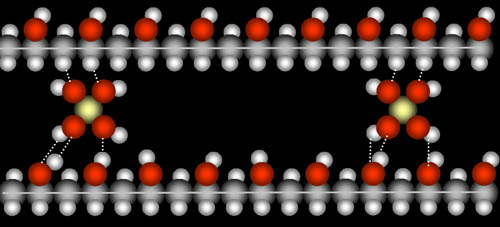
The salt used for crosslinking these polymer chains together is borax (sodium borate). Borate has one boron atom surrounded by four oxygen atoms, each with a hydrogen. Borate's hydrogen atoms are attracted to the oxygen atoms in the polymers (dotted lines). Likewise, borate's oxygen atoms are attracted to the hydrogen atoms in the polymers. This holds the chains of polymers together, which gives it more stiffness. The more borate molecules the more stiff the slime will be.

You will use the 250 mL beaker, the 100mL graduated cylinder, and the small plastic cup that came with your kit.

The polyvinyl alcohol powder you have will need to be dissolved in water and that takes some heating and a lot of stirring.
Some background information
I did this experiment three times in order to get it where I think you will have success. My first time was a failure. I followed the instructions from a book that gave measurements in teaspoons and tablespoons. It also said that hot tap water was all you needed to dissolve the polyvinyl alcohol. When this didn't work, I found another source on making slime which appeared more scientific. I followed those instructions and it worked fine.
Basically the instructions were to make up a 4% solution of polyvinyl alcohol and a 4% solution of borax. In medicine and chemistry a 4% solution is normally interpreted to mean 4 grams of the solid dissolved in every 100 milliliters of water.



For this experiment, I weighed the tube filled with polyvinyl alcohol. It measured 10.8 grams.
I then found the weight of the empty polyvinyl test tube. It was 6.0 grams (see below). Therefore, by subtracting the weight of the empty tube from the full tube of 10.8 grams, I found that you had 4.8 grams of polyvinyl alcohol in your kit.
But we could not put this in 100 mL of water, because it would not be a 4% solution. We needed more water because we have more than 4.0 grams; we had 4.8 grams. So here's the math. The fraction 4 grams over 100 mL is 4% solution. If we divide 4 by 100, we get 0.04. We need 4.8 to be divided by some volume that is also equal to 0.04. So we make an equation.
4 grams = 4.8 grams
100 mL ? volume
To solve for ? volume, we can do some cross multiplying.
4 grams x ? volume = 4.8 grams x 100 mL
Then divide both sides by 4 grams.
4 grams x ? volume = 4.8 grams
x 100 mL
4
grams 4
grams
The 4 grams cancel on the left and we end up with:
? volume = 120 mL
So we need to dissolve the 4.8 grams of polyvinyl alcohol in 120 mL of water in order to have the same concentration as 4 grams in 100 mL of water (4% solution).


The Borax tube with borax weighed 12.4 grams. The empty tube was the same as the empty polyvinyl alcohol tube (6.0 grams). So that means the borax tube must be holding 6.4 grams of borax. To make a 4% solution we need to dissolve the 6.4 grams in enough water to be equal to 4%. We use the same formula as before.
4 grams = 6.4 grams
100 mL ? volume.
4 grams x ? volume = 6.4 grams x 100 mL
Then divide both sides by 4 grams.
4 grams x ? volume = 6.4 grams
x 100 mL
4
grams 4
grams
The 4 grams cancel on the left and we end up with:
? volume = 160 mL
So we need to dissolve the whole tube of borax into 160 mL of water in order to get 4%. Our graduated cylinder only goes to 100mL, so we will first measure out 100mL followed by 60mL.





If you were like me, the polyvinyl alcohol doesn't dissolve very easily. This is where I learned about the different purity levels of polyvinyl alcohol.
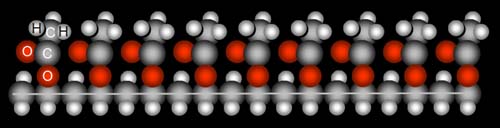

Apparently, polyvinyl alcohol is made from another polymer, polyvinyl acetate. See the two red oxygens, the two gray carbons, and three white hydrogens? That's acetate. When polyvinyl alcohol is made, these acetates are supposed to come off and be replaced by alcohol groups (one oxygen and one hydrogen). Often not all of these acetates come off. This can make the polyvinyl alcohol polymer not as water soluble as it would be if all acetates had come off.
The container that I got the polyvinyl alcohol from did not indicate the purity. So it's possible that it contained some residual polyvinyl acetate, which made it less water soluble. So that means we have to heat it.

To begin I turned the electric stove temperature to the second lowest temperature. If using a gas stove, turn it as low as you can. A microwave oven is another option (see below).





Keep stirring and heating until most of it is dissolved. It will take several minutes. You won't get every bit of it too dissolve, but try to get as much as you can to dissolve.

Turn off the heat.






So here's our 4% polyvinyl alcohol solution on the left and the 4% borax solution on the right.
 Take
a picture of at this point before you mix the two ingredients.
Take
a picture of at this point before you mix the two ingredients.






At first it seemed like a clump of Jello. I was a little disappointed.
But then...

It started flowing, behaving much different than Jello.

Drooling and stretching while feeling cold and slimey.

It slowly engulfs whatever it encounters.
What if it also had the power to take control over the brain...?

 (Take
some pictures of your slime, even if it didn't work. Also include a picture
where I can see your face. No, you don't have to be slimed.)
(Take
some pictures of your slime, even if it didn't work. Also include a picture
where I can see your face. No, you don't have to be slimed.)

Elmer's Glue-All contains two polymers, polyvinyl alcohol and polyvinyl acetate. The borax cross-links these together to make something like Silly Putty. The beauty of this approach is that it's fast. No time needed to dissolve the polyvinyl alcohol powder; however, this isn't the one I want you to make for this lab. I just mention it for something you may do in the future.
The school children that come to our college on Science day make "Silly Putty." This "slime" is not translucent like the type we made here with polyvinyl alcohol and borax.
Email the tap water temperature, plus the picture taken before you mixed the polymer and the borax and some pictures after you mixed them, plus at least one with your face visible.